United Kingdom Vision Systems for Blister Pack Pharma Inspection Market Expands- Körber, Syntegon, Cognex
Stringent UK regulations, serialization mandates, and AI-powered inspection technologies accelerate adoption across pharma packaging lines.
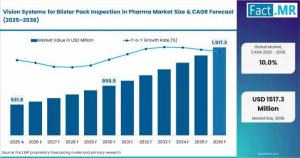
UNITED STATES, MD, UNITED STATES, April 21, 2026 /EINPresswire.com/ -- The United Kingdom vision systems for blister pack inspection in pharma market is entering a high-growth phase, driven by evolving compliance standards and the need for zero-defect pharmaceutical packaging. The global market is valued at USD 486.2 million in 2025 and is expected to reach USD 531.8 million in 2026, expanding to USD 1,382.4 million by 2036.Within this landscape, the UK market is projected to grow at a CAGR of 10.7% through 2036, supported by strong regulatory oversight and increasing deployment of automated inspection technologies across pharmaceutical manufacturing facilities. The transition toward AI-powered vision systems is reshaping quality assurance strategies, ensuring higher accuracy, traceability, and compliance.Get detailed market forecasts, competitive benchmarking, and pricing trends: https://www.factmr.com/connectus/sample?flag=S&rep_id=14788
Quick Stats
Global Market Size (2025): USD 486.2 Million
Global Market Size (2026): USD 531.8 Million
Global Forecast Value (2036): USD 1,382.4 Million
UK CAGR (2026–2036): 10.7%
Incremental Opportunity (Global): ~USD 850+ Million
Leading End-Use Segment: Pharma Manufacturers (46%)
Leading Technology: 2D Vision Systems (38%)
UK Market Position: Mature, compliance-driven growth
Key Players in UK: Körber AG, Syntegon Technology GmbH, Cognex Corporation, Antares Vision Group
Executive Insight for Decision Makers
The UK pharmaceutical sector is shifting toward fully validated, automated inspection ecosystems aligned with Medicines and Healthcare products Regulatory Agency (MHRA) requirements.
Strategic Priorities:
Deploy AI-enabled inspection systems for enhanced defect detection
Ensure compliance with serialization and traceability mandates
Integrate inspection systems with digital manufacturing platforms
Strengthen validation documentation capabilities
Risks of Delayed Adoption:
Regulatory non-compliance penalties
Increased recall risk due to packaging defects
Inefficiencies in high-speed production environments
Loss of competitive positioning in EU-aligned markets
Market Dynamics
Key Growth Drivers
Strict Regulatory Environment: MHRA guidelines enforce high packaging quality standards
Serialization Compliance: Ensuring traceability across supply chains
Automation in Pharma Manufacturing: Increasing reliance on high-speed packaging lines
AI and Machine Vision Adoption: Improved accuracy in defect detection
Key Restraints
High Implementation Costs: Advanced systems require significant capital investment
Complex System Integration: Challenges in retrofitting legacy production lines
Validation Requirements: Time-intensive approval processes
Emerging Trends
Adoption of AI-based anomaly detection systems
Integration with electronic batch record systems (EBR)
Growing use of high-resolution imaging and multispectral analysis
Expansion of smart manufacturing facilities in the UK pharma sector
Segment Analysis
Leading Segment: Pharmaceutical manufacturers dominate with 46% share, driven by compliance requirements
Technology Leader: 2D vision systems hold 38% share, offering reliable inspection for blister packaging
Fastest-Growing Segments
AI-powered inspection systems
Advanced imaging technologies
Key Applications
Tablet and capsule presence verification
Seal integrity inspection
Label and print verification
Contamination and defect detection
Strategic Insight:
Inspection systems are becoming integral to ensuring regulatory compliance and operational efficiency in UK pharmaceutical manufacturing.
Supply Chain Analysis (Critical Insight)
Value Chain Overview
Raw Material Suppliers:
Optical components, imaging sensors, lighting systems
Manufacturers:
Machine vision technology providers and inspection system developers
Distributors / Integrators:
Automation solution providers and packaging equipment integrators
End-Users:
UK-based pharmaceutical manufacturers and contract manufacturing organizations
“Who Supplies Whom”
Component suppliers → Vision system manufacturers
Vision system manufacturers → Packaging line integrators
Integrators → Pharmaceutical companies in the UK
Pharma companies → Healthcare providers and patients
Insight:
Close collaboration between system providers and UK pharma companies ensures seamless compliance with regulatory validation frameworks.
Pricing Trends
Pricing Structure:
Standard systems offer cost-effective solutions
AI-enabled systems command premium pricing
Key Influencing Factors:
Regulatory compliance requirements
System complexity and customization
Integration with packaging lines
Demand for high-speed inspection
Margin Insights:
Premium systems deliver higher margins due to advanced capabilities and compliance value.
Regional Analysis (United Kingdom Focus)
The UK represents a highly regulated and mature market, with growth driven by compliance and technological upgrades.
Key Growth Factors in the UK
Strong regulatory enforcement by MHRA
High adoption of automated packaging technologies
Presence of advanced pharmaceutical manufacturing infrastructure
Increasing investment in Industry 4.0 initiatives
UK vs Global Comparison
UK Market: Compliance-driven, technology-intensive
Emerging Markets: Expansion-driven, cost-sensitive
Global Leaders: Focus on innovation and AI integration
Competitive Landscape
Market Structure: Moderately consolidated
Key Players in the UK Market
Körber AG
Syntegon Technology GmbH
Cognex Corporation
Antares Vision Group
Teledyne DALSA Inc.
ISRA Vision GmbH
Competitive Strategies
Development of AI-driven inspection systems
Expansion of pharmaceutical-specific solutions
Strategic partnerships with UK pharma manufacturers
Focus on compliance and validation capabilities
Strategic Takeaways
For Manufacturers:
Upgrade to AI-enabled systems to meet evolving compliance standards
Focus on scalability and integration with digital systems
For Investors:
Target companies aligned with regulatory-driven demand
Focus on AI and automation innovation
For Distributors / Integrators:
Strengthen partnerships with pharmaceutical OEMs
Offer end-to-end compliance-focused solutions
Future Outlook
The UK market is set to evolve into a fully automated, compliance-driven inspection ecosystem.
Expansion of AI and deep learning technologies
Increased adoption of smart manufacturing systems
Growing emphasis on sustainability and waste reduction
Long-term growth will be driven by regulatory evolution, digital transformation, and pharmaceutical innovation.
Conclusion
The United Kingdom vision systems for blister pack inspection in pharma market is positioned as a benchmark for regulatory compliance and technological adoption.
Companies that invest in advanced inspection technologies and AI-driven solutions will lead the next phase of growth while ensuring operational excellence and compliance.
Why This Market Matters
In the UK, where regulatory standards are among the most stringent globally, vision inspection systems play a critical role in ensuring patient safety, product integrity, and supply chain transparency.
As pharmaceutical manufacturing continues to evolve, these systems will remain central to delivering quality, compliance, and competitive advantage.
Unlock 360° insights for strategic decision making and investment planning: https://www.factmr.com/checkout/14788
To View Related Report:
Robot Vision System Market https://www.factmr.com/report/robot-vision-system-market
Cleanroom Vision Panel Market https://www.factmr.com/report/cleanroom-vision-panel-market
Industrial Vision Gateways Market https://www.factmr.com/report/industrial-vision-gateways-market
Industrial Visioneers Market https://www.factmr.com/report/industrial-visioneers-market
S. N. Jha
Fact.MR
+ +1 628-251-1583
email us here
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.